Cryo-fixation is a technique used to immobilize the specimen at cryogenic temperatures. There are two techniques available in the BIF, plunge freezing and High Pressure Freezing (HPF).
A. Plunge-Freezing
Specimens are physically immobilized within ultra-thin vitrified (amorphous) ice layers. This technique is mainly used for freezing aqueous suspensions, such as macromolecules and cell suspension cultures.
Equipment:
Leica Vitrobot
B. High Pressure Freezing
Specimens are physically immobilized within milliseconds at high pressure and cryogenic temperatures. The sample must fit into a 3 mm or 6 mm diameter carrier. This technique is mainly used to preserve tissues that are 100 – 600 um thick.
Equipment:
Leica EM ICE
Advantages:
Complete cessation of all cellular processes (non-selective, within msec)
Problems caused by osmotic shock or inappropriate pH, often associated with
chemical fixation, are not present.
Disadvantages:
Formation of ice crystals could damage the ultrastructure.
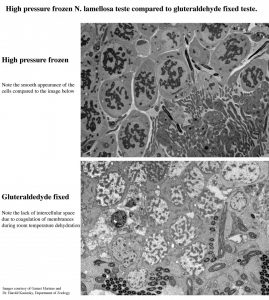
Comparison of High-pressure-freezing vs Chemical fixation:
| High Pressure Freezer | Chemical Fixation | |
| Fixation Time | Ultra rapid (msec) | Slow, simply by diffusion |
| Fixation Efficiency | High, non-selective | Variable
(A time lag in fixation between outer and inner tissue) |
| Fixation Artifacts | Formation of ice crystal | Many on TEM level
(Shrinkage, membrane distortion, aggregation of proteins/lipids) |
| Sample Size | Must fit into sample carrier | Variable |